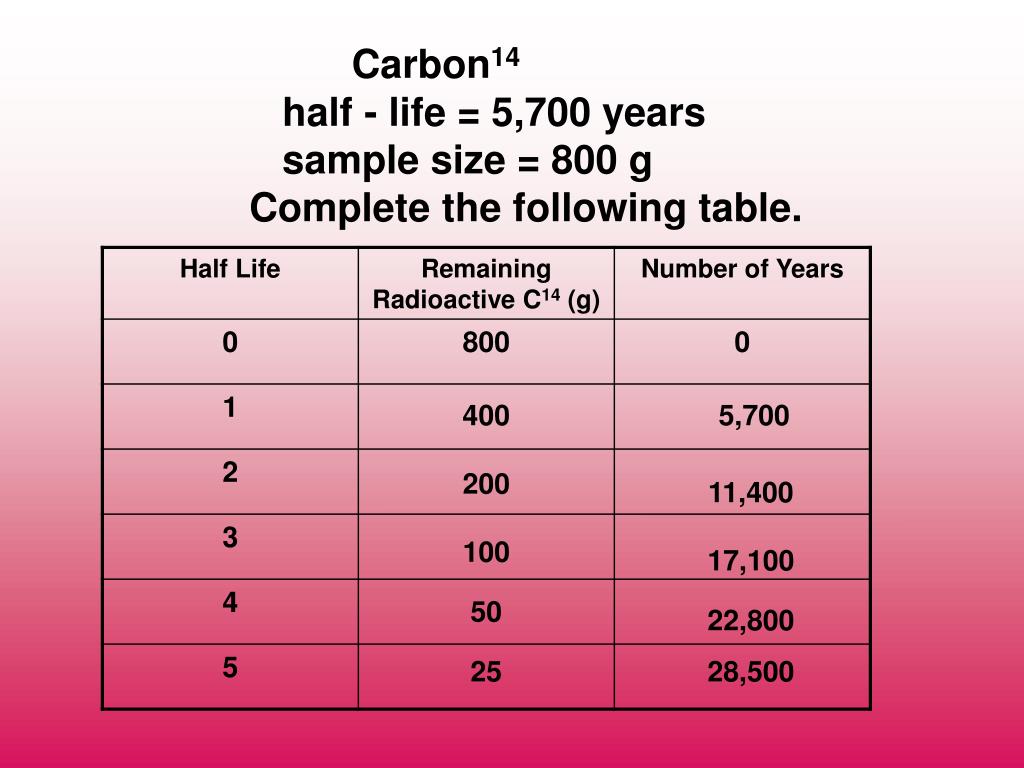
That is, 1 Bq = 1 decay/s.Īctivity R is often expressed in other units, such as decays per minute or decays per year. Carbon-14 decays into 14N through the process called Beta decay with a half-life of approximately 5,730 years. The SI unit for activity is one decay per second and is given the name becquerel (Bq) in honor of the discoverer of radioactivity. Where Δ N is the number of decays that occur in time Δ t. Therefore, the number of radioactive nuclei decreases from N to \frac\\ Further, half of that amount decays in the following half-life. Half of the remaining nuclei decay in the next half-life. The time in which half of the original number of nuclei decay is defined as the half-life, t 1/2.

Why use a term like half-life rather than lifetime? The answer can be found by examining Figure 1, which shows how the number of radioactive nuclei in a sample decreases with time. In two half-lives, what will remain is a quarter of the original substance because half of a half is a quarter, and so on. Remember that half-life is the amount of time it takes for half of the substance to decay into the daughter product. In this section we explore half-life and activity, the quantitative terms for lifetime and rate of decay. The half-life of carbon 14 is approximately 5,730 years. This means they have shorter lifetimes, producing a greater rate of decay. For example, radium and polonium, discovered by the Curies, decay faster than uranium. Carbon-14 decays with a halflife of about 5730 years by the emission of an. How many mg of a 20. Carbon dating is a variety of radioactive dating which is applicable. Manganese-56 decays by beta emission and has a half-life of 2.6 hours. How many grams of carbon-14 did the fossil originally contain 19. However, some nuclides decay faster than others. A 10,000 year old fossil currently contains 1.8g of carbon-14.

By the end of this section, you will be able to:
